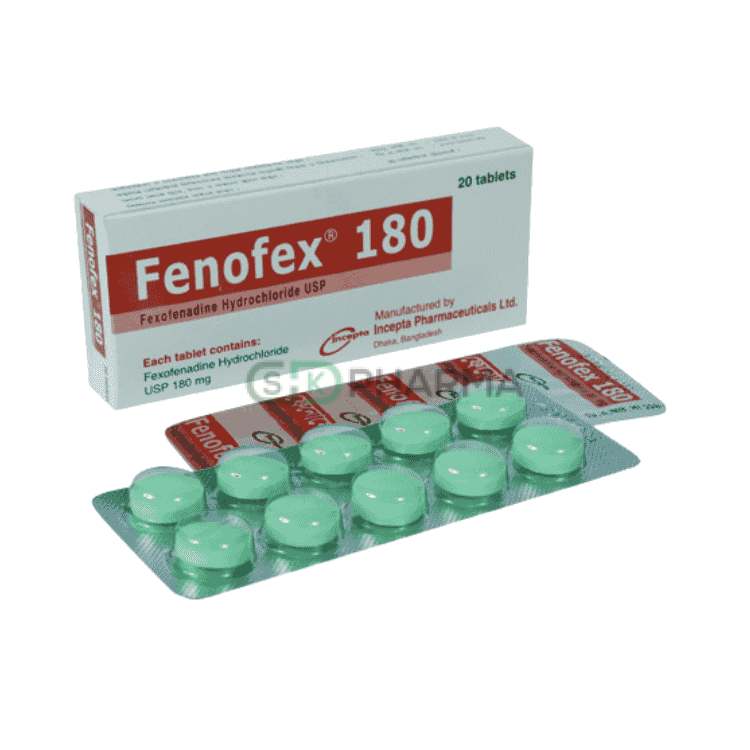
Fenofex Tablet 180 mg (Fexofenadine Hydrochloride)
-
৳3.33
৳3.50 -
৳5.64
৳6.00 -
৳8.46
৳9.00 -
৳32.90
৳35.00
Reviews & Ratings
Fenofex 180 mg is a non-sedating antihistamine containing Fexofenadine Hydrochloride, indicated for the effective management of allergic rhinitis and chronic idiopathic urticaria. It is specially formulated to provide long-lasting relief from allergy symptoms without causing drowsiness or impairing daily performance. Fenofex 180 mg helps improve work productivity, daily activities, and overall health-related quality of life in patients suffering from allergic conditions.
By selectively blocking peripheral H1 histamine receptors, Fenofex 180 mg prevents the development of common allergic symptoms while minimizing central nervous system effects. Due to its inability to cross the blood-brain barrier, it is considered safe for individuals who need to remain alert, such as drivers or machine operators.
রেজিস্টার্ড চিকিৎসকের পরামর্শ অনুযায়ী ঔষধ সেবন করুন
Composition
-
Active Ingredient: Fexofenadine Hydrochloride
-
Strength: 180 mg
-
Dosage Form: Tablet
Therapeutic Class
-
Non-sedating antihistamines
Indications
Fenofex 180 mg is indicated for use in adults and children aged 12 years and above for:
Allergic Rhinitis
-
Seasonal allergic rhinitis
-
Persistent allergic rhinitis
-
Sneezing
-
Rhinorrhea
-
Lacrimation
-
Itchy, red eyes
-
Itchy nose, palate, and throat
Chronic Idiopathic Urticaria
-
Reduction in wheals
-
Relief of pruritus
Pharmacology
Fenofex 180 mg is a selective peripheral H1-receptor antagonist. After oral administration, it is rapidly absorbed, reaching peak plasma concentrations within 2 to 3 hours. It does not readily cross the blood-brain barrier, which explains its minimal sedative effects. Fexofenadine does not undergo hepatic biotransformation, reducing the risk of metabolism-related drug interactions.
Mode of Action
Allergic reactions occur when histamine binds to H1 receptors following allergen exposure. Fenofex 180 mg acts as an inverse agonist at the H1 receptor, stabilizing it in an inactive form and preventing the release of pro-inflammatory mediators. This action effectively reduces itching, sneezing, watery eyes, and other allergy-related symptoms without anticholinergic or sedative effects.
Dosage and Administration
Allergic Rhinitis and Chronic Idiopathic Urticaria
Adults and children (≥12 years):
-
180 mg once daily
-
Alternative regimens: 120 mg once daily or 60 mg twice daily
Renal impairment:
-
Initial dose: 60 mg once daily
Dosage adjustments are required for pediatric patients and those with renal impairment, as advised by a healthcare professional.
Drug Interactions
-
Co-administration with erythromycin or ketoconazole may increase plasma levels of fexofenadine without clinical significance
-
No significant interaction with omeprazole
-
Antacids containing aluminum or magnesium hydroxide may reduce absorption; a 2-hour gap is recommended
Contraindications
-
Known hypersensitivity to fexofenadine or any component of the formulation
Side Effects
Fenofex 180 mg is generally well tolerated. Reported side effects include:
-
Headache
-
Fatigue
-
Dizziness
-
Nausea
-
Vertigo
Rare post-marketing effects include hypersensitivity reactions, palpitations, tachycardia, rash, urticaria, pruritus, anxiety, and sleep disturbances.
Pregnancy and Lactation
-
Pregnancy Category C (US FDA)
-
Use during pregnancy only if clearly necessary
-
Not recommended during breastfeeding due to excretion in breast milk
Precautions and Warnings
-
Use with caution in elderly patients and those with renal impairment
-
Patients with cardiovascular disease should be monitored for palpitations
-
Unlikely to impair driving or operating machinery, but individual response should be assessed
Overdose
Symptoms may include dry mouth, dizziness, fatigue, and somnolence. Supportive and symptomatic treatment is recommended. Hemodialysis is ineffective in removing fexofenadine.
Storage Conditions
-
Store in a dry place
-
Protect from heat and light
-
Keep out of the reach of children.
Related Products
Abeza Tablet 150 mg (Abemaciclib)
Ravu Capsule 100 mg (Ravuconazole)
Pink Pill Tablet 100 mg (Flibanserin)
Eracef Injection 1 gm/vial IV (Ceftriaxone Sodium)
Canaglif Tablet 100 mg (Canagliflozin Hemihydrate)
Vasoactin Tablet 500 mg (Citicoline Sodium)
Product Queries (0)
Login Or Registerto submit your questions to seller
Other Questions
No none asked to seller yet
-
৳3.33
৳3.50 -
৳5.64
৳6.00 -
৳8.46
৳9.00 -
৳32.90
৳35.00